![Facile, efficient and one-pot access to diverse new functionalized aminoalkyl and amidoalkyl naphthol scaffolds via green multicomponent reaction using triethylammonium hydrogen sulfate ([Et3NH][HSO4]) as an acidic ionic liquid under solvent-free ... Facile, efficient and one-pot access to diverse new functionalized aminoalkyl and amidoalkyl naphthol scaffolds via green multicomponent reaction using triethylammonium hydrogen sulfate ([Et3NH][HSO4]) as an acidic ionic liquid under solvent-free ...](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs11030-019-09945-4/MediaObjects/11030_2019_9945_Sch2_HTML.png)
Facile, efficient and one-pot access to diverse new functionalized aminoalkyl and amidoalkyl naphthol scaffolds via green multicomponent reaction using triethylammonium hydrogen sulfate ([Et3NH][HSO4]) as an acidic ionic liquid under solvent-free ...

Direct Conversion of Thiols to Sulfonyl Chlorides and Sulfonamides | The Journal of Organic Chemistry
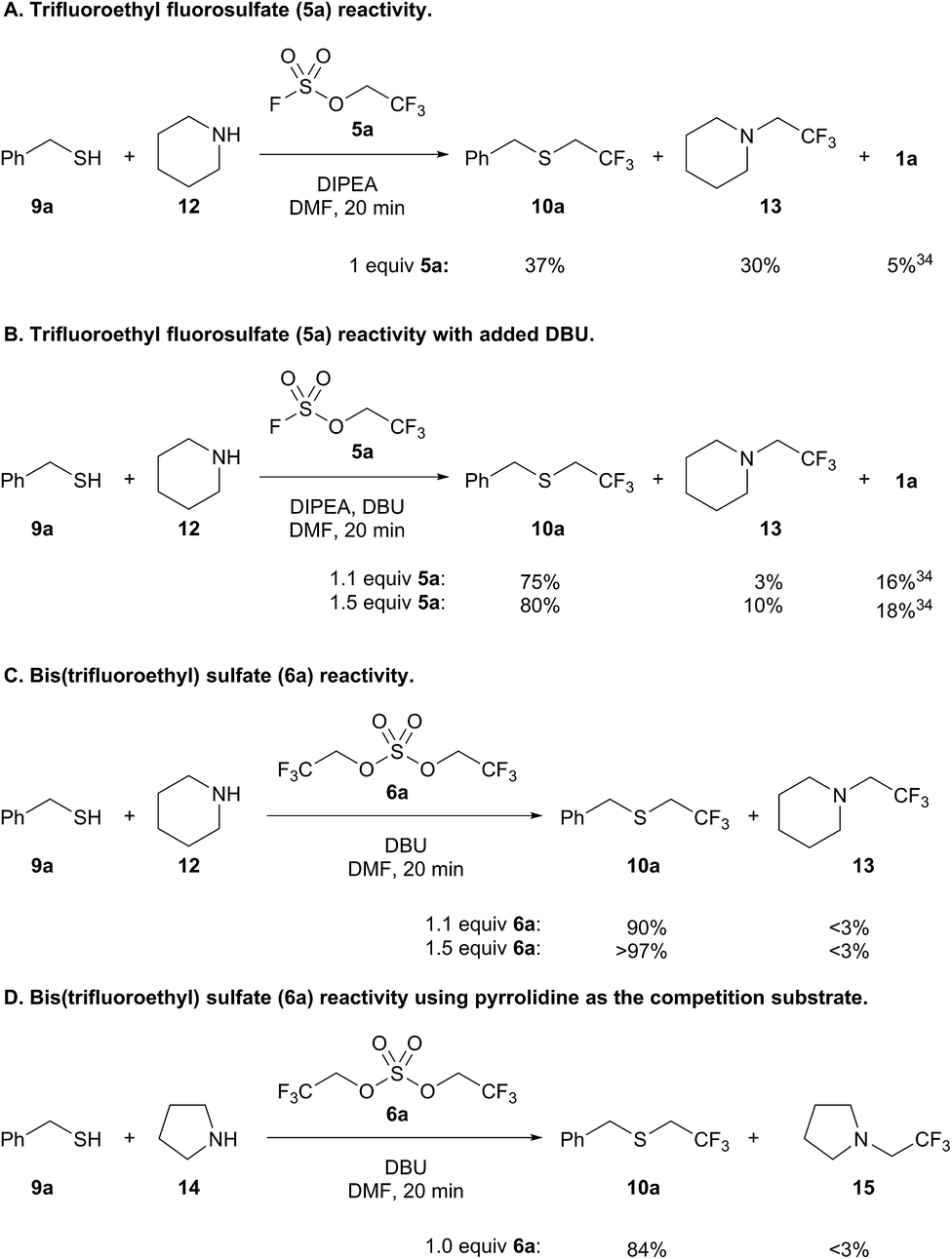
New sulfuryl fluoride-derived alkylating reagents for the 1,1-dihydrofluoroalkylation of thiols - Chemical Science (RSC Publishing) DOI:10.1039/C9SC03570B

LC/MS characterization of undesired products formed during iodoacetamide derivatization of sulfhydryl groups of peptides - Yang - 2007 - Journal of Mass Spectrometry - Wiley Online Library

LC/MS characterization of undesired products formed during iodoacetamide derivatization of sulfhydryl groups of peptides - Yang - 2007 - Journal of Mass Spectrometry - Wiley Online Library
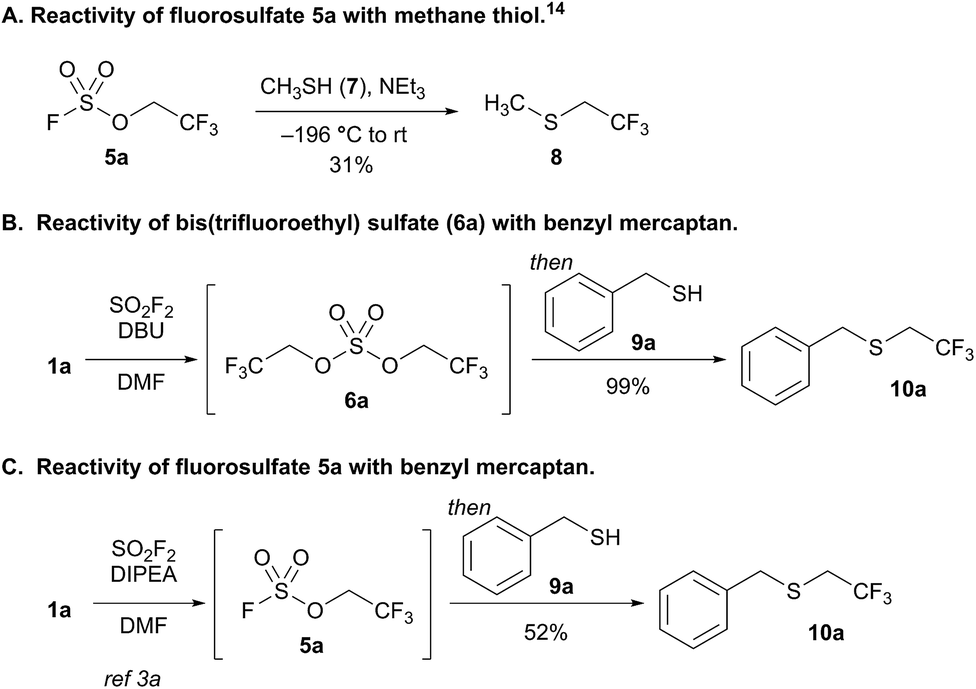
New sulfuryl fluoride-derived alkylating reagents for the 1,1-dihydrofluoroalkylation of thiols - Chemical Science (RSC Publishing) DOI:10.1039/C9SC03570B

TESIMAX připravuje inovace protichemických obleků a zásahových oděvů, jaké aktuálně nabízí? | POŽÁRY.cz - ohnisko žhavých zpráv | hasiči aktuálně

LC/MS characterization of undesired products formed during iodoacetamide derivatization of sulfhydryl groups of peptides - Yang - 2007 - Journal of Mass Spectrometry - Wiley Online Library
![Facile, efficient and one-pot access to diverse new functionalized aminoalkyl and amidoalkyl naphthol scaffolds via green multicomponent reaction using triethylammonium hydrogen sulfate ([Et3NH][HSO4]) as an acidic ionic liquid under solvent-free ... Facile, efficient and one-pot access to diverse new functionalized aminoalkyl and amidoalkyl naphthol scaffolds via green multicomponent reaction using triethylammonium hydrogen sulfate ([Et3NH][HSO4]) as an acidic ionic liquid under solvent-free ...](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs11030-019-09945-4/MediaObjects/11030_2019_9945_Sch1_HTML.png)
Facile, efficient and one-pot access to diverse new functionalized aminoalkyl and amidoalkyl naphthol scaffolds via green multicomponent reaction using triethylammonium hydrogen sulfate ([Et3NH][HSO4]) as an acidic ionic liquid under solvent-free ...

LC/MS characterization of undesired products formed during iodoacetamide derivatization of sulfhydryl groups of peptides - Yang - 2007 - Journal of Mass Spectrometry - Wiley Online Library

LC/MS characterization of undesired products formed during iodoacetamide derivatization of sulfhydryl groups of peptides - Yang - 2007 - Journal of Mass Spectrometry - Wiley Online Library

LC/MS characterization of undesired products formed during iodoacetamide derivatization of sulfhydryl groups of peptides - Yang - 2007 - Journal of Mass Spectrometry - Wiley Online Library